The current state and future directions of RNAi-based therapeutics; Nat Rev Drug Discov.
The current state and future directions of RNAi-based therapeutics by Ryan L. Setten, John J. Rossi and Si-ping Han
Setten RL, Rossi JJ, Han SP. The current state and future directions of RNAi-based therapeutics. Nat Rev Drug Discov. 2019 Mar 7. doi: 10.1038/s41573-019-0017-4.
Abstract
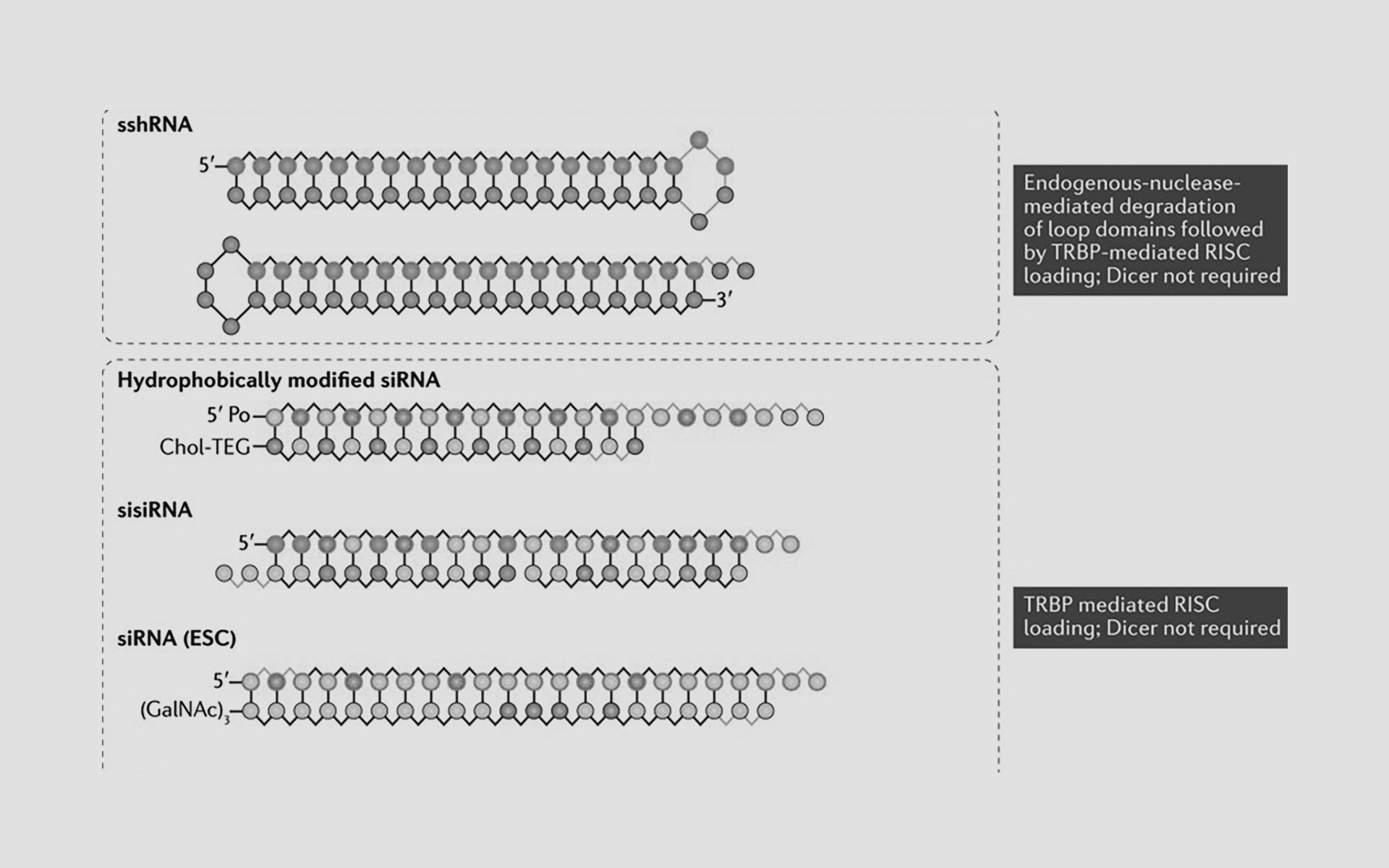
The RNA interference (RNAi) pathway regulates mRNA stability and translation in nearly all human cells. Small double-stranded RNA molecules can efficiently trigger RNAi silencing of specific genes, but their therapeutic use has faced numerous challenges involving safety and potency. However, August 2018 marked a new era for the field, with the US Food and Drug Administration approving patisiran, the first RNAi-based drug. In this Review, we discuss key advances in the design and development of RNAi drugs leading up to this landmark achievement, the state of the current clinical pipeline and prospects for future advances, including novel RNAi pathway agents utilizing mechanisms beyond post-translational RNAi silencing.
For PDF version, it should be solicited to Dr. Si-ping: sihan@coh.org
More information:

About The Author
Alex Lemus - President
Founding President of the Center for Biodefense and Global Infectious Diseases.